May 9, 2019
Scientists know that age and weight are risk factors in the development of cancer. That should mean that whales, which include some of the largest and longest-lived animals on Earth, have an outsized risk of developing cancer.
But they don’t. Instead, they are less likely to develop or die of this enigmatic disease. The same is true of elephants and dinosaurs’ living relatives, birds. Marc Tollis, an assistant professor in the School of Informatics, Computing, and Cyber Systems at Northern Arizona University, wants to know why.
Tollis led a team of scientists from Arizona State University, the University of Groningen in the Netherlands, the Center for Coastal Studies in Massachusetts and nine other institutions worldwide to study potential cancer suppression mechanisms in cetaceans, the mammalian group that includes whales, dolphins and porpoises. Their findings, which picked apart the genome of the humpback whale, as well as the genomes of nine other cetaceans, in order to determine how their cancer defenses are so effective, were published today in Molecular Biology and Evolution.
The study found that nature has beaten cancer in countless ways across the tree of life, Tollis said, and researchers can use that information to help find potential new targets for preventing cancer in humans, like using the whale version of a protein that can stop cell proliferation to develop drugs that shrink tumors in humans.
“Our goal is not only to get nature to inform us about better cancer therapies, but to give the public a new perspective of cancer,” Tollis said. “The fact that whales and elephants evolved to beat cancer, and that dinosaurs suffered from it as well, suggests that cancer has been a selective pressure across many millions of years of evolution, and it has always been with us. Our hope is that this may change people’s relationship with the disease, which can be painful and personal. It also helps provide even better appreciation for biodiversity. In our current sixth mass extinction, we need all the reasons for conservation that we can get.”
Why whales?
At its most basic, cancer occurs when body cells divide and mutate. Cell division is normal in living creatures, as are body cell, or somatic, mutations. Most of the time, somatic mutations are either harmless or the body is able to fix those mutations. When it doesn’t, that can lead to cancer.
Beyond that foundation, cancer gets much more complicated. For all of the research being done on all types of cancer and all types of organisms, there are still more questions than answers. However, age and body size are well-known risk factors in people. Tollis said one explanation for this is that cancer risk overall is a function of the number of cell divisions that occur over the lifetime of an organism.

“This is driven by somatic evolution—genetic changes that occur when body cells copy their genomes, divide and produce daughter cells,” he said. “The longer you live, the more cell divisions you have and the higher chance that a cancer-causing mutation will occur in the genome of the descendent cells. Similarly, larger individuals are made of more cells, which also increases the chance of cancer-causing mutations.”
Knowing this, it’s worth looking at whether whales, which live longer than most mammals on the planet and have a much higher percentage of body fat, are more likely to develop cancer. They aren’t. This phenomenon, known as Peto’s Paradox, is what Tollis and his team studied. How do whales defy these well-known risk factors?
Methods and conclusions
The team worked under a federal research permit to obtain skin samples from an adult female humpback whale off the coast of Massachusetts. This whale, named Salt, is well-known to researchers and whale watchers alike. She was cataloged by the Center for Coastal Studies in the mid-1970s and selected for this study because her life history is among the most well-documented of any individual humpback whale. The team sequenced and assembled Salt’s genome, which requires extracting DNA from skin tissue, fractioning it into smaller bits and putting it through a DNA sequencer that produces almost 2 billion short sequences. Then they put those sequences back together into a genome assembly that spans about 2.7 billion base pairs. (By contrast, the human genome is about 3.1 billion base pairs long.) They also sequenced RNA, which helps in the process of finding the precise coordinates where genes lie in the genome.
Tollis and his team then compared the humpback whale genome to those of other mammals including other ocean giants like the blue whale, fin whale, bowhead whale and sperm whale. They expected to see a lot of differences, but also a few similarities, particularly with the parts of the genome that perform important functions. They also looked for parts of the genome that had evolved to help whales adapt to their environment.
What they found was that some parts of the whale genome have evolved faster than in other mammals. These parts of the whale genome contain genes that control the cell cycle, cell proliferation and DNA repair, which are essential for normal cell function. In human cancers, many of these genes are mutated. The whale genome also evolved many duplications in tumor suppressor genes.
“Nature is showing us that these changes to cancer genes are compatible with life. The next questions are, which of these changes is preventing cancer, and can we translate those discoveries into preventing cancer in humans?” said Carlo Maley, a cancer evolutionary biologist from ASU’s Biodesign Institute and one of the two senior authors, who started this project in 2011 with Per Palsbøll, a marine mammal conservation geneticist from the Groningen Institute for Evolutionary Life Sciences at University of Groningen, in collaboration with Jooke Robbins from the Center for Coastal Studies.
“This suggests that whales are unique among mammals in that in order to evolve their gigantic sizes, these important ‘housekeeping’ genes, that are evolutionarily conserved and normally prevent cancer, had to keep up in order to maintain the species’ fitness,” Tollis said. “We also found that despite these cancer-related parts of whale genomes evolving faster than other mammals, on average whales have accumulated far fewer DNA mutations in their genomes over time compared to other mammals, which suggests they have slower mutation rates.”
This slow rate of change may also limit whales’ exposure to cancer-causing somatic mutations.
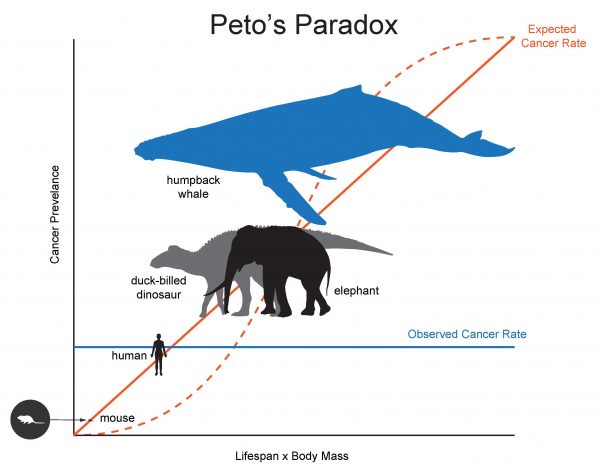
This study builds on research that the ASU team has done finding lower rates of cancer in elephants, another mammal that is large and can live a long time but doesn’t have much in common genetically with whales. Both species arrived at the same result—cancer suppression—through different mechanisms. The same is likely true of extinct giant dinosaurs; although scientists have found evidence of cancer in dinosaur fossils, this group of organisms includes the largest animals ever to walk the earth and thus they probably had efficient cancer suppression as well.
Next steps for Tollis and his team will be to better understand the cancer suppression phenotype using experiments with whale cell lines, which will provide important functional validation of the team’s genomic results. That will be the first step in generating whale-derived human cancer drugs. They also are looking at other animals, such as bats, tortoises and crocodilians, to see how these varied long-lived species suppress cancer. The humpback whale genome also will serve as the basis for studies by other team members on whale mutation rates and other adaptations taking advantage of a collection of the 9,000-plus DNA samples collected from free-ranging humpback whales curated by Palsbøll’s team.
This research is supported by the Arizona Cancer Evolution Center, which is funded through a five-year grant from the National Institutes of Health, with additional funding by the University of Groningen.

Heidi Toth | NAU Communications
(928) 523-8737 | heidi.toth@nau.edu



